If your browser does not re-direct you, please click here to go to our new page.
Using hPSC derived cortical neurons; we are modeling human cortical networks in vitro. This provides a novel platform towards understanding how human cortical networks develop, as well as their mature properties.
Furthermore this approach allows us to investigate the dysfunction of cortical circuits in human neurodevelopmental disorders such as Down syndrome.
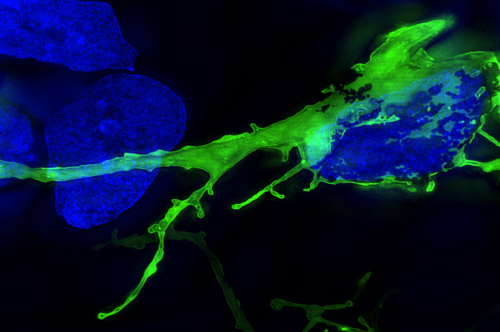
The initiation of AD has been linked to the accumulation of the amyloid-beta peptide and the formation of plaques. A key question is to determine a link between its initiation and progression.
Aβ has been shown to interact with several signalling pathways that regulate the phosphorylation of the microtubule associated protein tau, that is hyperphosphorylated and forms neurofibrillary tangles in Alzheimers. Interestingly, mutant forms of tau have been shown to spread trans-synaptically in a defined spatio-temporal pattern in the AD brain.
To this effect, we are characterising these secondary neuropathological changes in tau and investigating its spread in patient specific cortical neurons.
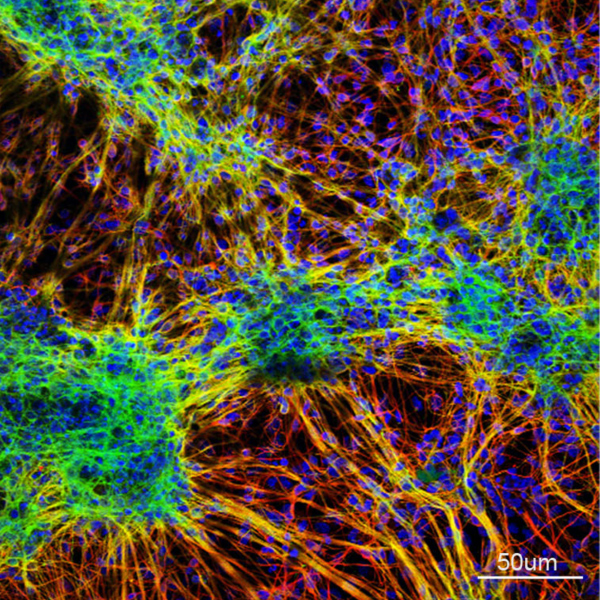
How can stem cells give rise to the full complexity of the cerebral cortex?
One project combines lineage tracing with quantitative and computational approaches to understand the rules that govern neural stem cell behaviour in development. We are particularly interested in the contribution of altered stem cell dynamics to neurodevelopmental disorders.
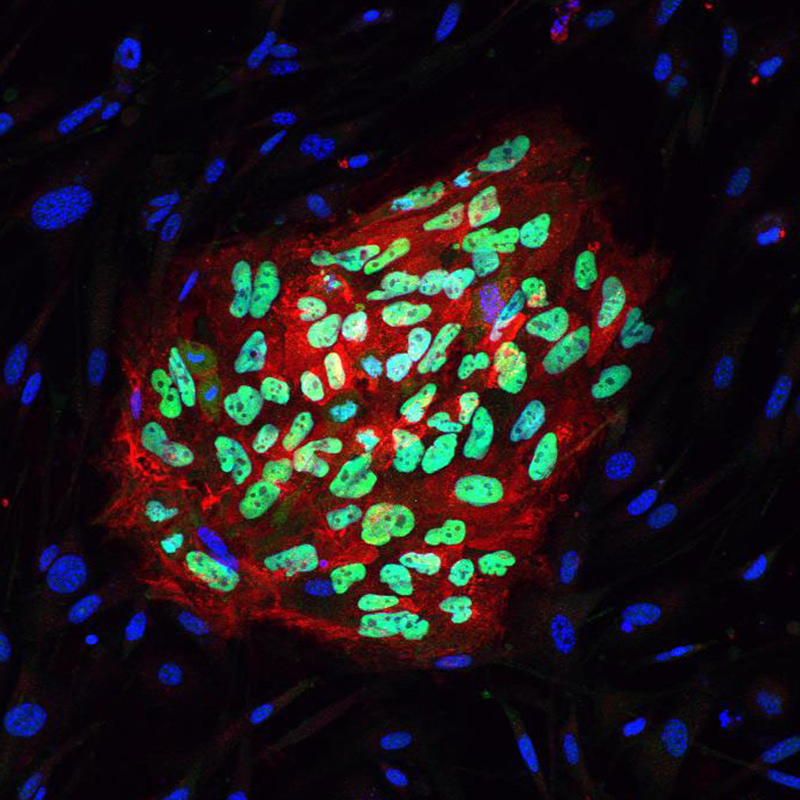
The combination of IPS cells and the ability to differentiate these into specific cell types provides a powerful tool to human study disease in vitro.
In order to maximise this potential we are using TALENs and the CRISPR system to manipulate our IPS lines and investigate the role of gene copy number or SNPs on development and disease.
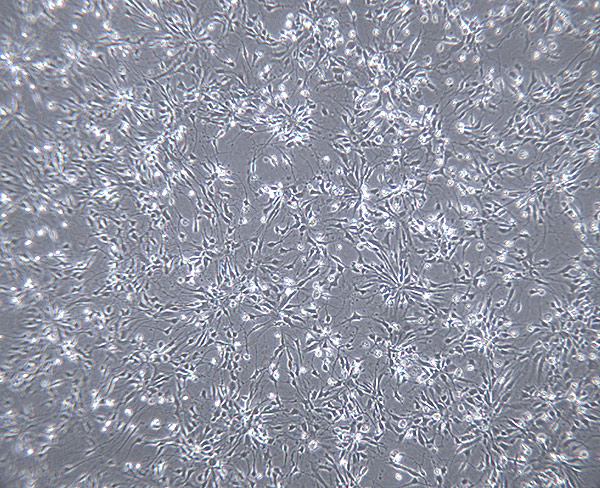
The directed differentiation protocol established by the lab efficiently produces human cortical progenitor cells that give rise to the full complement of excitatory neurons over time.
We are interested in building upon our existing culture protocols by manipulating developmental signalling pathways to specify neuronal progenitors with distinct identities across the anterior-posterior and dorso-ventral axis of the CNS. The aim is to apply these studies to the in vitro modelling of human CNS development in addition to the cellular specificity of neurodevelopmental disorders and neurodegenerative diseases.
In evolution, the cerebral cortex has enlarged dramatically along the human lineage both in terms of size and number of constitutive neurons. It is widely believed that this expansion is the basis of high cognitive functions of human brains.
By applying our in vitro model of corticogenesis to non-human primates, we are able to directly compare cortical development of humans with that of our closest relative species. This should allow us to identify cellular and genetic mechanisms underlying the evolution of the human cerebral cortex.

Building on the Down Syndrome iPS cell model for Alzheimer's », we have developed a system to screen compounds for potential to alter pathology in disease cell lines.
Using a combination of high-throughput cell culture and screening assays, we aim to monitor the effect of treatment with pharmacologically active molecules on known AD markers. This provides us with a platform for novel drug identification and screening.